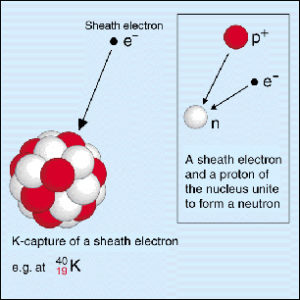
During beta decay, the proton in the nucleus is transformed into a neutron and vice versa. What happens in beta decay?īeta decay is a radioactive decay in which a beta ray is emitted from an atomic nucleus. Gamma rays are produced during gamma decay of an excited nucleus. They are the most energetic of all electromagnetic waves. 
Gamma rays are electromagnetic waves that carry photons of energy called gamma particles. The number of protons (and neutrons) in the nucleus does not change in this process, so the parent and daughter atoms are the same chemical element.ĭuring gamma decay, an excited nucleus emits a particle. 3-6, a nucleus changes from a higher energy state to a lower energy state through the emission of electromagnetic radiation (photons). Which particle has the highest penetrating power?Īnswer and Explanation: Gamma particles have the highest penetrating power among the given types of radiation. What causes beta decay?īeta decay occurs when an atom has either too many protons or too many neutrons in its nucleus. Inverse beta decay, commonly abbreviated to IBD, is a nuclear reaction involving an electron antineutrino scattering off a proton, creating a positron and a neutron. According to quantum theory, it is impossible to predict when a particular atom will decay, regardless of how long the atom has existed. random) process at the level of single atoms. How do you calculate radioactive decay?Ĭalculations Using the First Order Rate Equation: r = k Since the rate of radioactive decay is first order we can say: r = k1, where r is a measurement of the rate of decay, k is the first order rate constant for the isotope, and N is the amount of radioisotope at the moment when the rate is measured. What is radioactive decay constant?ĭecay constant, proportionality between the size of a population of radioactive atoms and the rate at which the population decreases because of radioactive decay. Alpha particles have a relatively large mass and a positive charge. This ejected particle is known as an alpha particle and is simply a helium nucleus. What happens in alpha decay?Īlpha decay is a nuclear decay process where an unstable nucleus changes to another element by shooting out a particle composed of two protons and two neutrons. This is usually in the form of alpha particles (Helium nuclei), beta particles (electrons or positrons), or gamma rays (high energy photons). Radioactive decay is the random process in which a nucleus loses energy by emitting radiation. 24 What can stop gamma rays? What is radioactive decay A level physics?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
